Top>Opinion>A New Energy System to Mitigate Global Warming
 Index
Index

Hiroyuki Hatano【profile】
A New Energy System to Mitigate Global Warming
Hiroyuki Hatano
Professor, Faculty of Science and Engineering, Chuo University
Areas of specification: Reaction Engineering and Process Systems, Fluidization, and Desiccant Air Conditioning
Chemical-looping system
Researchers around the world are trying to develop clean and highly efficient energy systems to solve problems faced by modern societies, such as global warming and energy resource depletion. Such methods as using lattice oxygen of metal oxides to combust fossil fuels or gasifying solid fuels have been studied for years; however, a new combustion system free of the energy penalty of carbon capture was first developed by a research group at the Tokyo Institute of Technology in Japan. The system was named “chemical-looping combustion.” Although carbon capture was considered impractical at first, this social climate has changed since nuclear power plants were suspended after the Great East Japan Earthquake and the power plant disaster in Fukushima. While an increasing number of new coal thermal power plants are being planned as a cheaper and less geopolitically risky option, large-scale chemical-looping technology development projects are taking off in Western countries. Even the results of a one-MWth pilot plant study were reported at the International Conference on Chemical Looping held every two years[1]. At other international conferences on energy and global warming, the number of presentations on chemical-looping systems is rapidly increasing. In Japan as well, coal chemical-looping combustion is being actively studied, mainly by foundations and private corporations, and a further advanced research study is to be launched in FY2015.
Overview of chemical-looping combustion
As shown in Eq. (1), chemical-looping combustion uses the heat released during oxidation of metal M by oxygen in the air into metal oxides MO. Then, as shown in Eq. (2), a hydrocarbon fuel is used to reduce MO into M. With these two reactions combined to remove M and MO, a general equation for hydrocarbon combustion can be derived, as shown in Eq. (3). However, if the reactions of Eqs. (1) and (2) are separately performed in the air reactor (AR) and fuel reactor (FR), respectively, carbon dioxide can be captured by cooling the exhaust gas of Eq. (2) down to room temperature since this reaction produces only carbon dioxide and water vapor. Thus, this process can condense oxygen for combustion by using a solid as a mediator. Since Q1 (the amount of heat released) is generally much larger than Q2 (the amount of heat absorbed), it can be safely assumed that the chemical-looping combustion system works as a high-temperature chemical heat pump. A schematic diagram is shown in Fig. 1.
AR: (2n + m/2) M + (n + m/4) O2 → (2n + m/2) MO + Q1 (exothermic) (1)
FR: (2n + m/2) MO + CnHm → M + nCO2 + (m/2) H2O - Q2 (endothermic) (2)
Combustion: CnHm + (n + m/4) O2 → nCO2 + (m/2) H2O + Q3 (= Q1 + Q2) (3)
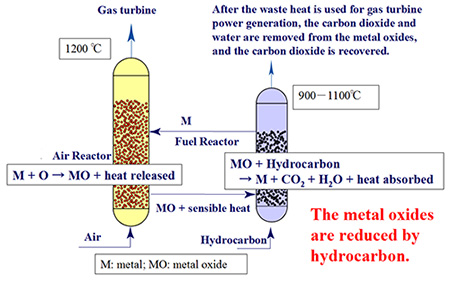
Fig. 1. Chemical-looping combustion system using heat from aerial oxidation of iron
(With a smaller amount of lattice oxygen, this system can be applied to the gasification and reforming of hydrocarbon for hydrogen production)
Overview of chemical-looping gasification
The principle of chemical-looping gasification has been known for years, and numerous relevant studies have been conducted. For gasification, the reduction reaction of Eq. (2) should be stopped halfway to obtain CO, CO2, and H2. It is also possible to produce pure hydrogen with the metal-water reaction, as shown in Eq. (4).
HG (Hydrogen Generator): M + H2O → MO + H2 (4)
In chemical-looping combustion, which handles particles at high temperatures, it is still difficult to use solid fuels such as coal. As illustrated in Fig. 2 (schematic diagram)[2], the hydrogen production system with chemical-looping gasification is a three-phase system where particles are circulated through the hydrogen generator (HG), AR, and FR.
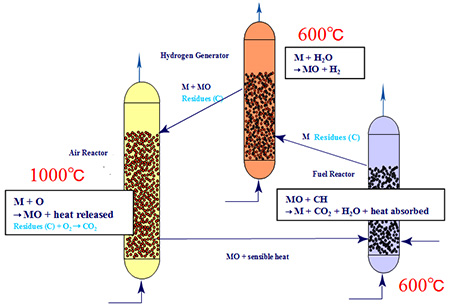
Fig. 2: Three-phase particle circulating fluidized bed hydrogen production system with CO2 recovery
New energy systems tailored for individual communities
The chemical-looping system is applicable for generating biomass power using grass and wood chips. In this case, like other energy systems, it is still difficult to operate large generators and solve problems such as low efficiency and high cost. Nevertheless, the system can be unexpectedly useful if it is used as a mechanism to revive communities or preserve biodiversity. In fact, the author found the potential of a community based on biomass energy systems while discussing the basic policies of his Department of Integrated Science and Engineering for Sustainable Society, which covers such a wide range of disciplines that the students are to take a general seminar course in their third year of undergraduate studies and a cross-disciplinary course when the graduate course is developed.
For example, people have continued to carry out a burning practice in the Watarase Retarding Basin, with a total area of 3,300 hectares, to protect endangered species. However, it can be more beneficial to mow the field instead of burning it and recruit volunteers to do it. Based on the assumption that a hectare of land produces 10 tons of plant biomass per year in dry weight, it is estimated that 33,000 tons of dry biofuel can be produced every year in the basin. This is almost equivalent to the amount required to fuel an incinerator with a capacity of 100 tons per day. This, however, generates only several thousand kW of power, which means that the energy efficiency is as low as 15 percent. The actual power generation capacity, or the optimal capacity of the power generator, would be even smaller than that considering the workforce available for mowing and collecting biomass.
On the other hand, the energy efficiency can be increased to 70 to 80 percent by using the biomass as a heat source. Where can this energy be used? It should be possible to use it for small-scale pyrolysis gasification to recover carbide, tar, and combustion ash for soil amelioration, water purification, and agrochemical production. Meanwhile, the waste heat and carbon dioxide of the exhaust gas can be utilized to warm greenhouses and for other agricultural purposes in the basin area. Moreover, health promotion can be achieved by establishing a mechanism to make use of local volunteers to mow grass and harvest biomass.
If it is difficult to use the waste heat for agricultural purposes, it can be used for air conditioning. In this case, the development of biomass energy can be associated with the establishment of a community hub for people to meet each other, such as a multi-generational community center with chauffeur services for young and elderly people. This idea can be applied to activate urban areas as well. It is, for example, advisable to establish a multi-generational community center in the vicinity of a waste incineration facility so that the area can attract people. This may reduce daytime energy consumption at individual homes while creating a stable demand for the waste heat of the incineration facility. Moreover, social costs can be reduced if measures are taken such as enhancing the vitality and health of the elderly, reducing the number of infants on waiting lists for childcare services, and improving after-school childcare services.
Further studies are needed to assess these issues more precisely. For example, the above-mentioned seminar courses of study can be utilized for this purpose.
This paper introduces not only energy systems being used at present but also the possible applications of waste heat to be developed for the future. Both have many problems to overcome. The author is planning to continue studying these matters to establish a sustainable society.
References:
- Hiroyuki Hatano
Professor, Faculty of Science and Engineering, Chuo University
Areas of specification: Reaction Engineering and Process Systems, Fluidization, and Desiccant Air Conditioning - Professor, Faculty of Science and Engineering, Chuo University (from April 2013 – up to date)
Research Group Leader/Senior Researcher/Chief Senior Researcher, National Institute of Advanced Industrial Science and Technology (from April 2001 to March 2013)
Part-time Lecturer, Department of Mechanical Engineering, Faculty of Science and Technology, Tokyo University of Science (from April 2010 up to date)
Senior Researcher/Laboratory Chief, National Institute for Resources and Environment, reorganized from National Research Institute for Pollution and Resources, Ministry of International Trade and Industry (from July 1990 to March 2001)
Postdoctoral research fellow, West Virginia University (from October 1987 to September 1988)
Assistant, Production Facility Division, Chemical Resource Laboratory, Tokyo Institute of Technology (from April 1983 to June 1990)
- Research Activities as a Member of Research Fellowship for Young Scientists (DC1), Japan Society for the Promotion of Science (JSPS) Shuma Tsurumi
- Important Factors for Innovation in Payment Services Nobuhiko Sugiura
- Beyond the Concepts of Fellow Citizens and Foreigners— To Achieve SDGs Goal 10 “Reduce Inequality Within and Among Countries” Rika Lee
- Diary of Struggles in Cambodia Fumie Fukuoka
- How Can We Measure Learning Ability?
—Analysis of a Competency Self-Assessment Questionnaire— Yu Saito / Yoko Neha - The Making of the Movie Kirakira Megane