Top>Research>Material that shrinks when heated
 Index
Index

Kengo Oka [profile]
Education Course
Material that shrinks when heated
Kengo Oka
Assistant Professor, Faculty of Science and Engineering, Chuo University
Areas of Specialization: Solid State Chemistry and Solid State Property
The familiar thermal expansion phenomenon
The thermal expansion phenomenon, where volume expands on heating, is a common quality seen in almost all material. Instruments utilizing the thermal expansion phenomenon, such as alcohol thermometers that measure temperature with the expansion of liquid sealed in glass tubes and bimetals made by joining differing thermal expansion coefficients used in temperature sensors, are useful in our daily lives. However, thermal expansion is also a troubling phenomenon that brings about various problems. For example, when boiling water is suddenly poured into a glass tumbler, it cracks. This is due to the stress caused by thermal expansion when the inside of the cup suddenly becomes hot with the boiling water. Openings in rail track and bridge joints are also for prevention of thermal expansion problems. Thermal expansion is a highly familiar phenomenon, but do we really know how large thermal expansion is?
Problems brought about by thermal expansion
The thermal expansion phenomenon should appear on a daily basis, but there are barely any chances to notice changes in length through thermal expansion with the naked eye. The magnitude of thermal expansion is, as a linear thermal expansion coefficient (proportion in change of length for each 1℃ change in temperature), several ten ppm /℃ for most material. For example, when a 10 cm iron rod (linear thermal expansion coefficient of 12 ppm/℃) rises in temperature by 1℃, it expands 1.2 μm. You may think that this is so small a change that you couldn’t notice it with your eyes. But in precision machines such as automobile engines which are operated in high temperature, where the change in length is hundreds of times of this value, they must be designed precisely with calculations taking account of deformations from thermal expansion. Also, in recent nanotechnology, thermal expansion is a serious issue. For example, the process steps in the fabrication of CPU for the latest computers have been miniaturized to under 20 nm = 0.02 μm. In industries such as the semiconductor industry, which demand such fine positioning, a 1℃ change in temperature can cause a fatal misalignment.
Solving thermal expansion problems
So what do we need to do to prevent problems arising from thermal expansion? There are two approaches we can take. The first is a method that maintains a set temperature. In principle, if temperatures do not change, then problems related to thermal expansion will not appear. However, in order to maintain a set temperature, equipments and a huge amount of energy for advanced air-conditioning is required. The other approach is to have the material not susceptible to thermal expansion itself. There will be many people who will ask the question, “Sure, if material does not expand on heating, we can solve the problem, but is it really possible?” But there is a strange material that makes this possible.
Making non-thermal expansion material with negative thermal expansion
Making non-thermal expansion material – the key to that is the strange phenomenon that turns conventional wisdom on its head, where material shrinks on heating. Most material expands on heating, but there are, albeit a small number, strange material that shrinks on heating. In contrast to material that expands when exposed to heat possessing thermal expansion coefficients with positive values, because the coefficient values of material that shrinks on heating are negative, they are called negative thermal expansion material. If positive and negative thermal expansion cancel each other out with combination of this two materials, we can keep thermal expansion at zero. Low-thermal expansion glass with negative thermal expansion material (LiAlSiO4etc) as a filler is actually commercially available and is used for cooktops and superheat resistant glass containers.
You may be thinking, if there is material that shrinks when heated and we actually can control thermal expansion, then hasn’t the problem been solved? However, there is one big problem. That is, the thermal expansion coefficient of negative thermal expansion material isn’t very large. Because the thermal expansion coefficient of glass material is originally low (linear thermal expansion coefficient:<10 ppm/℃), controlling thermal expansion is comparatively easy, but this is not the case for metals and resins where thermal expansion is large. Whereas the highest linear thermal expansion coefficient of commercially available manganese nitride is -40 ppm/℃, metals and resins have large linear thermal expansion coefficient values of 10-30 ppm/℃ and 50-200 ppm/℃ respectively. In order to maintain the properties of the structure material, the filler amount of negative thermal expansion material must be small. For that purpose, we have studied a giant negative thermal expansion material that surpasses conventional material.
Giant negative thermal expansion through electrical charge transfer
What do we need to do to achieve giant negative thermal expansion? There, our research group focused on the material where volume changed from large to small when pressure is applied. The metal oxide BiNiO3, a compound synthesized using high pressure apparatus that is used for producing diamonds, takes on a crystalline structure called perovskites. This material takes the strange valence state of Bi3+0.5Bi5+0.5Ni2+O3 under ambient pressure owing to the exotic nature of bismuth ion. By applying pressure, the charge transfer between Bi to Ni occurs and the valence state changes to Bi3+Ni3+O3 above 3.5 GPa. Because the valence of Ni increases from 2 to 3, the Ni-O bonding contracts, resulting in a 2.5% shrinkage in volume. We thought, if we apply this large volume contraction, we could create negative thermal expansion material surpassing conventional material.
The contraction in the volume of BiNiO3 lies in its close relationship with the electronic state. By chemically changing the electronic state, we should be able to control the phase transition that causes this volume contraction. There, we tried substituting Bi with other trivalent ions. This substitution should stabilize the valence state of Bi3+Ni3+O3 and volume contraction should occur under ambient pressure. By substituting Bi with La (lanthanum), volume contraction occurs on heating without applying pressure. During this phase transition, the large and small phases coexist with changing their proportion gradually. As a result, the average volume continuously reduced as the temperature rose. A giant negative thermal expansion of -82 ppm/℃ was observed. This is a negative thermal expansion double that of conventional material, and comparable with resin material.
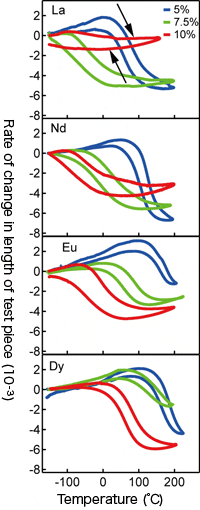
Thermal expansion behavior of the samples with bismuth substituted with lanthanoid (La, Nd, Eu, Dy). Negative thermal expansion tuning can take place by changing the substituting ion and amount.

Change in valence state when bismuth (Bi) is partially replaced with lanthanoid.
Our research group conducted neutron diffraction and X-ray absorption spectroscopy experiments, and founded that the charge transfer between Bi and Ni occurred at lower temperatures by increasing the amount of substituted La. In other words, it shows that it is possible to chemically control negative thermal expansion. We also founded that the negative thermal expansion behaviors for BiNiO3 based materials shows dependence to the chemical compositions for Ln = La, Nd, Eu, Dy. It is possible to tune the magnitudes and temperature ranges of negative thermal expansion of this materials for specific purposes. This is a huge advantage when thinking of practical application. Furthermore, we discovered that the sample substituted Ni with Fe shows giant thermal expansion of more than -150 ppm/℃ in near room temperature.
Zero thermal expansion resin material
So, is it possible to create a zero thermal expansion resin material using small amounts of the newly discovered material? There, we actually tried to make a zero thermal expansion composite material by applying BiNi0.85Fe0.15O3 (linear thermal expansion coefficient of -187 ppm/℃) for epoxy resin (linear thermal expansion coefficient of 80 ppm/℃). The result was, with an added amount of 18 vol%, this composite showed zero thermal expansion at between 30℃ and 60℃ and this temperature range can be controlled by tuning negative thermal expansion property of this material. we succeeded in controlling thermal expansion at a zero level.
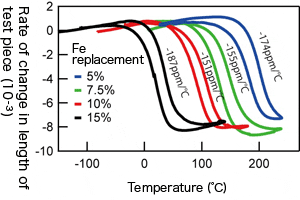
Thermal expansion behavior of test piece where Ni is substituted with Fe. By changing the amount of substituted Fe, the temperature range of negative thermal expansion can be controlled.
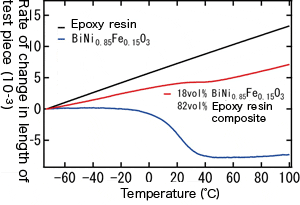
Thermal expansion behaviors of the epoxy resin, BiNi0.85Fe0.15O3 where15 % of Ni has been substituted by Fe, and the composite material with 82 vol% of epoxy resin and 18 vol% BiNi0.85Fe0.15O3. Thermal expansion of the composite material was almost zero between 30℃and 60℃.
Conclusion
Negative thermal expansion material in BiNiO3 based material exhibits outstanding properties that can tune the temperature range of negative thermal expansion and have giant negative thermal expansion several times more than conventional material. The advantage of the large negative thermal expansion means we can even realize zero thermal expansion even for resin materials. Because thermal expansion is a common property in all material, we hope that thermal expansion control technology will be used in all areas, not only nanotechnology such as semiconductor device manufacturing and optical communication.
Results of this research have been obtained from the joint research of the research group led by the Tokyo Institute of Technology Material and Structures Laboratory Professor Masaki Azuma.
-
Kengo Oka
Assistant Professor, Faculty of Science and Engineering, Chuo University
Areas of Specialization: Solid State Chemistry and Solid State Property - Kengo Oka was born in Kyoto in 1983. He completed his doctorate in Department of Chemistry at the Graduate School of Science, Kyoto University in 2010. He holds a Ph.D. (Science). After working as a Japan Society for the Promotion of Science Research Fellow for Young Scientists (Institute for Solid State Physics, the University of Tokyo), and Specially Appointed Research Professor at the Tokyo Institute of Technology Material and Structures Laboratory from 2010, he entered his current position in 2014. His current field of research is synthesis and physical property research of new functional material including Pb and Bi.
- Research Activities as a Member of Research Fellowship for Young Scientists (DC1), Japan Society for the Promotion of Science (JSPS) Shuma Tsurumi
- Important Factors for Innovation in Payment Services Nobuhiko Sugiura
- Beyond the Concepts of Fellow Citizens and Foreigners— To Achieve SDGs Goal 10 “Reduce Inequality Within and Among Countries” Rika Lee
- Diary of Struggles in Cambodia Fumie Fukuoka
- How Can We Measure Learning Ability?
—Analysis of a Competency Self-Assessment Questionnaire— Yu Saito / Yoko Neha - The Making of the Movie Kirakira Megane