Top>Research>New hybrid hydrogen carrier
 Index
Index

Chang Ho-Chol [profile]
Education Course
New hybrid hydrogen carrier
Chang Ho-Chol
Professor of Chemistry, Faculty of Science and Engineering, Chuo University
1. Hydrogen continues to draw attention
It goes without saying that energy issues are one of the most important problems faced by mankind today. In Japan, following the Great East Japan Earthquake, there has been increased public attention and debate regarding renewable energy such as solar power, wind power and geothermal power. Electricity can be generated from renewable energy. However, many forms of renewable energy are often controlled by time and are influenced by geographical conditions. Therefore, in addition to generating power, it is extremely important to convert the generated electricity into a chemical substance (chemical energy) for storage and transportation. (It would be even better to directly produce chemical substance from renewable energy.) Hydrogen (H2) has attracted attention as a chemical substance which is capable of storing such energy. As a fuel, hydrogen does not evolute CO2 and other greenhouse gases when burned. It is a clean energy which only forms water (see formula below).
H2 + 1/2O2 → H2O
Hydrogen can also be generated through a method known as steam reforming for water and methane (CH4), which is a natural gas that can be obtained directly. Another method for generating hydrogen is electrolysis of water. Compared to such technology for generating hydrogen, many serious issues remain in technology for storing hydrogen. Hydrogen exists as a gas at normal temperatures and normal pressure. Currently, hydrogen is cooled to its boiling point of -253℃. The resulting liquid is then stored in a large and heavy high-pressure-resistant tank for shipping.
2. Hydrogen carrier
In order to realize a hydrogen society which uses hydrogen as society’s energy infrastructure, it is essential to develop technology for efficiently storing hydrogen in a substance which can hold hydrogen (known as a hydrogen carrier), safely shipping the hydrogen to locations where it is needed, and effectively extracting only the required amount at the required time. The ideal hydrogen carrier must possess high hydrogen content, be environmentally friendly and be very easy to handle. Such hydrogen carriers have been the subject of research since long ago. In Japan, researchers led by Emeritus Professor Masaru Ichikawa of Hokkaido University have worked hard to develop organic hydride from organic material.[1] For example, methylcyclohexane shown in Figure 1 is replaced with toluene while evoluting trimolecular hydrogen. In other words, methylcyclohexane acts as a hydrogen carrier. Currently, ammonia and alcohol are also viable candidates in addition to methylcyclohexane. However, an endothermic reaction is required to extract hydrogen from these carriers, thus creating the need for high temperatures. Furthermore, these carriers require expensive catalysts such as platinum. Therefore, while it is also important to improve upon existing materials and technology, there is demand for developing a completely new hydrogen carrier and inexpensive catalysts, as well as for new methods of extracting hydrogen.
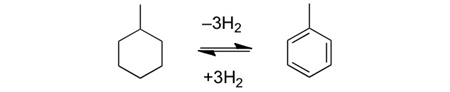
Figure 1: Organic hydride composed of methylcyclohexane/toluene. Requires high temperatures and a catalyst such as platinum.
3. Our idea
Conversely, for many years, we have conducted research which focuses on the skeleton shown in Figure 2. As shown in the figure, the characteristic of this skeleton is that the aromatic skeleton which contains oxygen and sulfur atoms (represented by “E” in Figure 2) can be bonded with a variety of metal ions. Interestingly, bonding with metal facilitates the entrance/exit of electrons in this organic skeleton, something which is normally quite difficult. For example, if a single electron is removed from the group of electrons (known as a catecholate) shown at the left of Figure 2, the structure oxidizes into a semiquinonato (middle of Figure 2). When one electron oxidation is performed for this semiquinonato, it oxidizes to the final form of a benzoquinone (right of Figure 2). Accordingly, it is possible to remove/insert two electrons from the overall structure. Moreover, although semiquinonato is normally an extremely reactive radical, bonding with metal ions creates a stable compound which can be handled even in the open atmosphere.
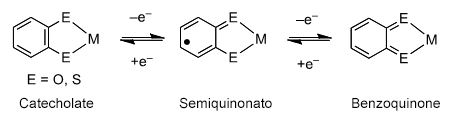
Figure 2: Complex unit formed from redox-active ligands from which two electrons can be transferred
We thought that if it were possible to remove/insert the negatively charged electron (e-) from this skeleton, and then, if it would also be possible to introduce the positively charged proton (H+) which is the opposite of the negative charge, we could remove/insert electrons and protons. If it were possible to remove two electrons and two protons and then bond them together…that’s right! The result would be hydrogen (H2) (see formula below).
2H+ + 2e- → H2
My research partners and I targeted the skeleton o-phenylenediamine (opda) (left of Figure 3). When two electrons and two protons are removed from this skeleton, it changes into a skeleton known as benzoquinone diimine (right of Figure 3). However, at that time, there was no report of hydrogen having been generated from opda. Indeed, there was no clear report for the bonding of opda and metal ions. One reason for this is that opda and metal ions react instantaneously in the atmosphere when bonded and isolation is extremely difficult. The only useful information which we possessed was a report detailing how hydrogen radicals (H·) were generated when an aromatic amine similar to opda was irradiated with light. Although a white powder was obtained when mixing opda and Fe ion (Ⅱ), this powder is oxidized instantaneously when exposed to the atmosphere and absolutely no hydrogen was generated.

Figure 3: Complex unit from which two electrons and two protons can be transferred
4. Hydrogen carrier activated by light
However one day, researcher Dr. Matsumoto discovered that gas was generated when the mixture of opda and Fe ion (Ⅱ) was irradiated with light at an ambient temperature. When using gas chromatography to analyze that gas, we confirmed that the gas was hydrogen. As the result of repeated experiments and analyses, we discovered that the white powder obtained by mixing the two substances was a complex molecule in which opda and Fe ion (Ⅱ) have bonded. This complex is known as a tris-o-phenylenediamine complex (see Figure 4). Through our experiments, we were also able to clarify that the electrons and protons required to create the evoluted hydrogen originated from opda.
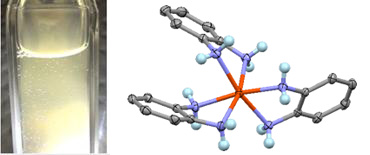
Figure 4. Generation of photo-hydrogen (left) and the complex molecule which is a hydrogen carrier (right)
Next, we set out to prove that this molecule was truly a catalyst which could store hydrogen and evolute that hydrogen when exposed to light. To establish proof, we conducted an optical irradiation experiment with coexistence of hydroquinone (left of Figure 5), a reagent which can supply both electrons and protons. We confirmed that the amount of hydrogen generated increased dramatically, compared to when there was no coexistence of hydroquinone. We also demonstrated that large amounts of hydrogen were not generated with hydroquinone alone. This proved that the molecule was definitely a catalyst.
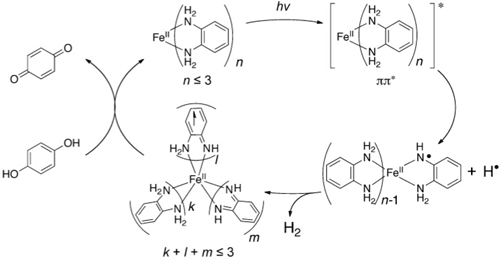
Figure 5: Proposed mechanism for catalytic hydrogen generation via light
The hydrogen carrier which we discovered has several characteristics. First, it operates at an ambient temperature. The majority of organic hydrides require temperature of 100℃ or higher in order to extract hydrogen. You could say that we used the method of optical irradiation to overcome this obstacle. The ability to generate hydrogen via optical irradiation creates promise of using solar power in the future. Second, it operates using iron, a very inexpensive metal. Until now, expensive noble metals such as platinum and iridium were used as catalysts. This creates great promise from the perspective of cost. The final characteristic is that the hydrogen carrier which we discovered is a hybrid carrier which is activated through the combination of an organic skeleton and inexpensive metal. Although further and more detailed research is necessary, it may be possible to improve functionality through conversion to similar organic skeletons or to apply the discovery to lightweight and inexpensive metals other than iron.
5. Conclusion
After all, we have only reached the stepping-off point for our discovery. There are still countless issues which much be addressed. We shall continue to actively conduct research for realizing the great promise, dreams and hope of developing an unprecedented hydrogen carrier and contributing to the solution of energy issues. Fortunately, our research results were featured on the cover of the Journal of the American Chemical Society issued by American Chemical Society and have been transmitted throughout the world (Figure 6). [2][3] In the future, we expect to further clarify the mechanism of our discovery, improve functionality, and realize application to diverse photochemical reactions. Finally, I would like to thank Dr. Tsuyoshi Matsumoto (currently active at the International Institute for Carbon-Neutral Energy Research, Kyushu University) and the research group of Hokkaido University for their contributions to this research.

Figure 6: Featured on the cover of the Journal of the American Chemical Society
References
- ^ All About Hydrogen Energy—Hydrogen Society and Hydrogen Business (written by Masaru Ichikawa; Ohmsha, Ltd.)
- ^ T. Matsumoto, H.-C. Chang, M. Wakizaka, S. Ueno, A. Kobayashi, A. Nakayama,
T. Taketsugu, M. Kato, J. Am. Chem. Soc., 2013, 135, 8646-8654. - ^ Homepage of Chang Laboratory http://www.chem.chuo-u.ac.jp/~chang/
- Chang Ho-Chol
Professor of Chemistry, Faculty of Science and Engineering, Chuo University - Born in Kyoto Prefecture in 1973. Graduated from the Faculty of Science, Korea University in 1995. Completed the Masterfs Program in the Graduate School of Science, Tokyo Metropolitan University in 1998. Completed the Doctoral Program in the Graduate School of Engineering, Kyoto University in 2001. Holds a PhD in engineering. Served as a Research Associate/Assistant Professor in the Graduate School of Engineering, Kyoto University and an Associate Professor in the Faculty of Science, Hokkaido University before assuming his current position in April 2013. Also serves as a JST PRESTO Researcher (concurrent position), etc.
- Research Activities as a Member of Research Fellowship for Young Scientists (DC1), Japan Society for the Promotion of Science (JSPS) Shuma Tsurumi
- Important Factors for Innovation in Payment Services Nobuhiko Sugiura
- Beyond the Concepts of Fellow Citizens and Foreigners— To Achieve SDGs Goal 10 “Reduce Inequality Within and Among Countries” Rika Lee
- Diary of Struggles in Cambodia Fumie Fukuoka
- How Can We Measure Learning Ability?
—Analysis of a Competency Self-Assessment Questionnaire— Yu Saito / Yoko Neha - The Making of the Movie Kirakira Megane